Where each single covalent bond represents a sigma bond, the one double bond represents a sigma bond and a pi bond*. A pi bond between two atoms is formed only in addition to a sigma bond. Each of the bonds formed during the hybridization process are direct, sigma bonds, where the last bond, formed simply by the nearness of the two atoms to each other, is a pi bond. The reason is that the overlapping of atomic orbitals can take place to a greater extent during the formation of a sigma bond, whereas overlapping of orbitals occurs to a smaller extent during the formation of a pi bond. That, my readers, is the difference between a sigma and a pi bond. Now, I wonder where we discussed two different kinds of bonds before… Although this one is different, isn’t it? This bond wasn’t formed through hybridization, but more as a consequence of two atoms looking for an additional electron that the other one had. This is contrasted by sigma bonds which form bonding orbitals directly between the nuclei of the bonding atoms, resulting in greater overlap and a strong. If you guessed “they form a bond of their own”, you would be correct. Before going any further into molecular orbital theory, make sure you know exactly what sigma and pi bonds are: Sigma bond: When. I’ll give you one guess as to what happens between the 2p orbitals of both carbons.
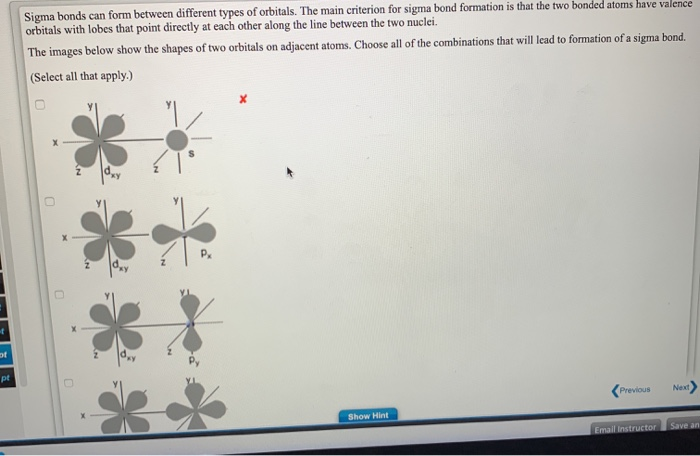
They can occur between any kind of atomic orbitals the only requirement is that the atomic orbital overlap happens directly. A sigma bond is the strongest type of covalent bond in which the atomic orbitals directly overlap between the nuclei of two atoms. Similarly, a triple bond contains one sigma and two pi bonds. Sigma () and Pi () bonds form in covalent substances when atomic orbitals overlap. A single bond always contains one sigma bond and a double bond contains one sigma and one pi bond. Pi bonds are created by the lateral overlap of two atomic orbitals, whereas sigma.
#Sigma bonds plus
This means that the other carbon that has already formed a bond with our carbon of interest also has a 2p orbital free. one pi-type bond Triple Bond one sigma-type bond plus two pi-type bonds Example: Complete. Sigma bond is the first bond between two atoms whereas pi bonds constitute all of the remaining bonds present in a compound. The overlapping of atomic orbitals is the main difference between these bonds. Remember that there is no central atom here - the molecule is symmetrical what happens to one carbon happens to the other.

